Cardanit BPMN editor becomes the key solution for companies’ merger


Organization: EUC Premium
Design challenge: Streamline the merger process of two healthcare companies
Benefits:
Identify and efficiently describe the companies' processesIntegrate into a unified environmentCollaborate with all stakeholders
How BPMN and Cardanit collaborative features helped the merge of two healthcare companies
Business Process Model and Notation is quite a simple standard. People who are not familiar with BPMN elements may also need a description of the diagram of the model implemented during the process analysis. This is why business consultants and analysts tend to share a more readable ‘user manual’, which Cardanit produces at the click of a button, when presenting the results of their work.
Learn how Cardanit helped Michal Breda, Process Modeling Specialist at EUC Premium in the Czech Republic, efficiently streamline the merger process of two healthcare companies. Michal and his team managed to smoothly draw hundreds of BPMN workflows in a very short time. At the same time, using Cardanit collaboration features, and most of all the automated report, Michal’s team shared their results with the different stakeholders from the two companies. All the processes were clearly understood and the Cardanit report became the starting point of their internal Quality Management process document.
The deadline for the merger was 1 April 2021 and Cardanit enabled EUC Premium to:
- Identify all the business processes needed for the merger
- Quickly and efficiently describe the processes of both companies
- Easily integrate them into a unified environment
- Work cooperatively in the description and implementation of the processes
- Comprehensively transfer the entire process system of both companies to a new structure
The ContextBusiness process documentation
In the middle of 2019, the management of EUC decided to describe all the processes in its subsidiary EUC Premium. About 180 processes have been identified as part of this project, most of which have been described by the end of 2019.
The ChallengeBusiness process integration
In November, a merger of EUC Premium with another subsidiary, Canadian Medical was announced. It was necessary to describe the business processes in this company and to start their integration as part of the merger. At the turn of the year, 80% of Canadian Medical processes were described.
From January to the end of March 2020, they worked on integrating the processes of both companies into a unified environment. Integration was split into several stages depending on the possibilities of integration of information systems. Starting from April 2020, the teams of the two companies have worked together, with an expected completion date of the integration of all the processes by 1 April 2021.
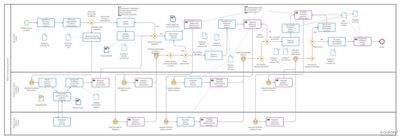
The SolutionCardanit BPMN editor
In Michal Breda’s words, “because we were able to quickly and efficiently describe the processes of both companies with the BPMN methodology in the Cardanit environment, it helped to manage the integration of the processes of both companies. These processes were partly different depending on the various information systems, and the graphical models helped all teams understand the processes of the other side and design a common integrated functional system.”
I've found very useful the collaboration features, the share functionality and the automated report.
The BenefitsCardanit collaboration features and automatic report
“During the merge phase, but also during my daily activities, I’ve found very useful the collaboration features, the share functionality and the automated report.”, Michal continues describing his process modeling with Cardanit.
“We usually set up a meeting in which we describe the process and we start designing the model on a whiteboard. I then draw it in Cardanit and after agreement of the diagram by the cooperating departments I generate the automated report. At that point, my colleagues involved in the project add comments under each activity, so we can complete the workflow based on their suggestions. We then transform the Cardanit report into our Quality Management process document and publish it on our intranet for common use.”
Michal and his team are now heading towards the completion of the merger process of EUC Premium with Canadian Medical efficiently drawing their processes with the help of Cardanit collaboration features.
Further reading
BPMN becomes corporate practice in healthcare business process management
Six ingredients for efficient process mapping
BPMN Explained: 7 questions every business analyst asks about BPMN



Michal Breda is Process Modeling Specialist at EUC and Canadian Medical, Czech Republic. Michal studied system engineering at CTU Prague and has an MHA from the Advance Healthcare Management Institute in Prague. He is an expert in the management of medical institutions. In his work, he applies extensive experience from long-term activities as CIO, Chief Operating Officer and Operational-Technical Deputy of several large medical institutions and private clinics. Since June 2019, he has been cooperating with EUC a.s. and Canadian Medical s.r.o. in the field of process analysis and modeling.
Michal Breda is Process Modeling Specialist at EUC and Canadian Medical, Czech Republic. Michal studied system engineering at CTU Prague and has an MHA from the Advance Healthcare Management Institute in Prague. He is an expert in the management of medical institutions. In his work, he applies extensive experience from long-term activities as CIO, Chief Operating Officer and Operational-Technical Deputy of several large medical institutions and private clinics. Since June 2019, he has been cooperating with EUC a.s. and Canadian Medical s.r.o. in the field of process analysis and modeling.
Learn what key features and ingredients your BPMN editor should have to ensure a smooth and efficient business process mapping experience.
Key ingredients for efficient process mapping
Learn what key features and ingredients your BPMN editor should have to ensure a smooth and efficient business process mapping experience.
Key ingredients for efficient process mapping
Learn what key features and ingredients your BPMN editor should have to ensure a smooth and efficient business process mapping experience.
